© Christian Kielmann
© Christian Kielmann
Willkommen beim AK Funktionsmaterialien an der TU Berlin
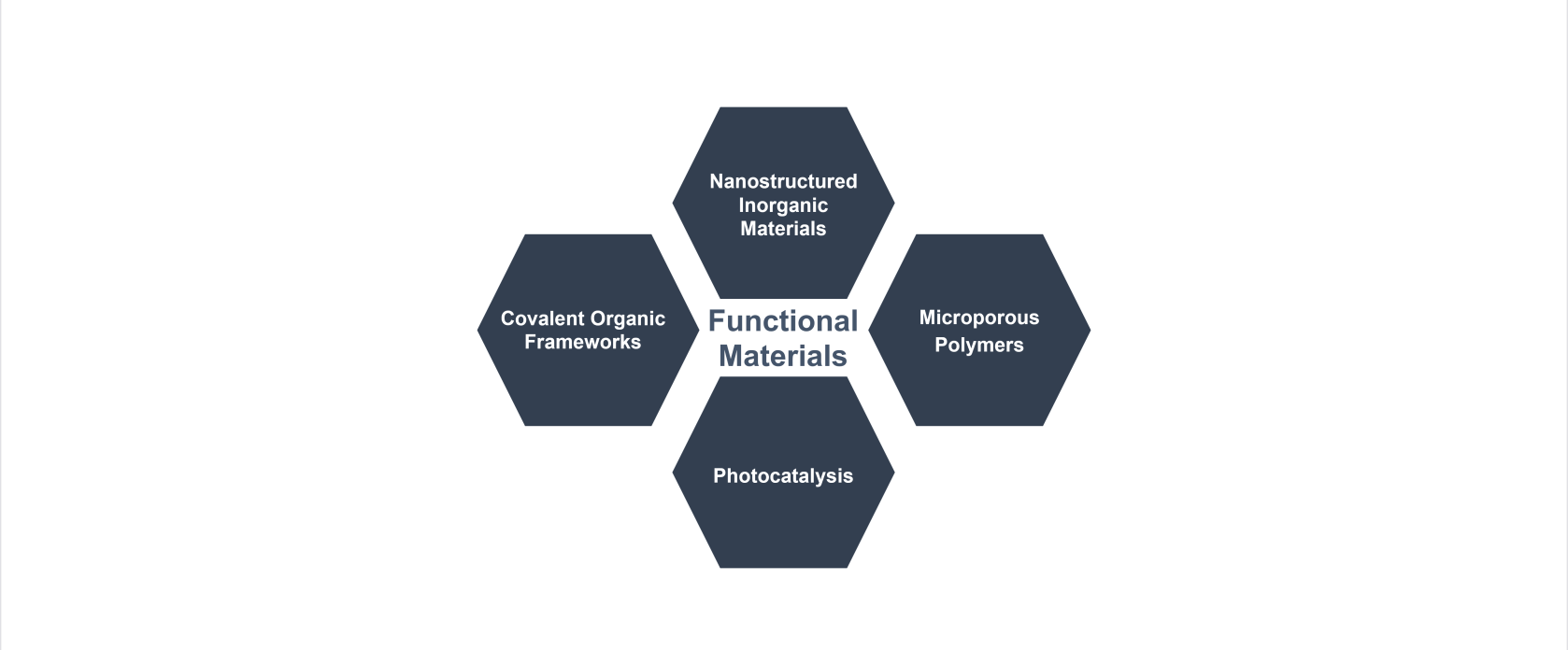
Unser Hauptinteresse gilt der Synthese von Materialien, deren Struktur und Eigenschaften zielgerichtet für eine bestimmte Anwendung eingestellt werden (Funktionsmaterialien).
Viele Materialien weisen dabei nur dann die gewünschte Funktion auf, wenn sie in einer bestimmten Struktur vorliegen oder in dieser organisiert sind. Die Einführung von Porosität in ein Material und damit die Maximierung seiner Oberfläche, ist dabei ein oft gewählter Ansatz, um die Funktionalität eines Materials zu verbessern oder eine neue Funktion zu generieren. Die chemische Zusammensetzung und Struktur solcher Materialien soll dabei für jede spezielle Anwendung angepasst bzw. eingestellt werden.
Wir beschäftigen uns daher sowohl mit anorganischen- und organischen Materialien, als auch anorganisch-organischen Hybridmaterialien
| Gebäude | BA |
|---|---|
| Raum | 216 |
| Adresse | Hardenbergstr. 40 10623 Berlin |
| Fakultät | II |
| Institut für | Chemie |
| Sekretariat | BA2 |